Establish an end-to-end process for complaints and defects
Your quality processes are fragmented, documenting defects is tedious, and rectifying them is inefficient? You are not alone: Many companies find it difficult to record complaints and defects contextually and resolve them in compliance with standards. This often results in high ramp-up costs for series production and dissatisfied customers. CONTACT Elements provides a solution for these challenges. With its functions for Quality Issue Management, you establish an end-to-end process that links all relevant data, standardizes workflows, and creates a sustainable solution archive.
Your benefits
- Increase product quality, reduce series ramp-up costs, and ensure compliance
- Establish integrated processes from error detection and recording to resolution and a solution archive
- Create a basis for continuous improvement

Increase product quality, reduce costs, and ensure compliance
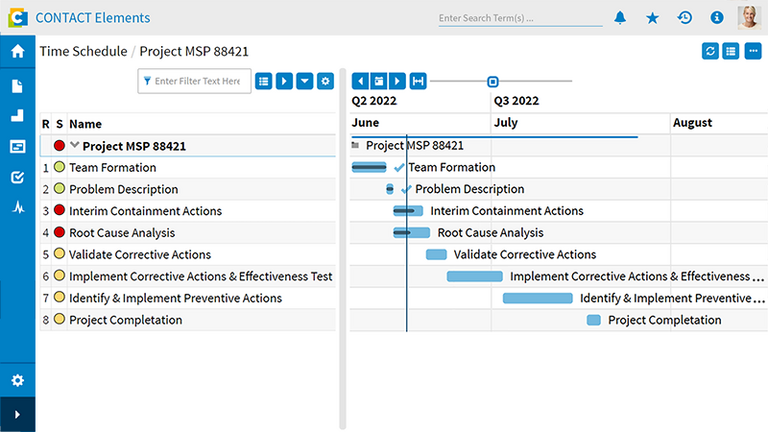
With CONTACT Elements Quality Issue Management, you handle quality defects with system support throughout the entire product lifecycle: from initial inspection through production ramp-up to ongoing production. This always occurs within the relevant product or project context. The solution ensures full conformity with important quality standards such as EN ISO 9001 and ISO 16949 through seamless documentation and traceability of every defect recording and resolution. Thanks to targeted defect resolution, you benefit from higher customer satisfaction, improved product quality, reduced production ramp-up costs, and faster production maturity.
Increase product quality, reduce costs, and ensure compliance
With CONTACT Elements Quality Issue Management, you handle quality defects with system support throughout the entire product lifecycle: from initial inspection through production ramp-up to ongoing production. This always occurs within the relevant product or project context. The solution ensures full conformity with important quality standards such as EN ISO 9001 and ISO 16949 through seamless documentation and traceability of every defect recording and resolution. Thanks to targeted defect resolution, you benefit from higher customer satisfaction, improved product quality, reduced production ramp-up costs, and faster production maturity.
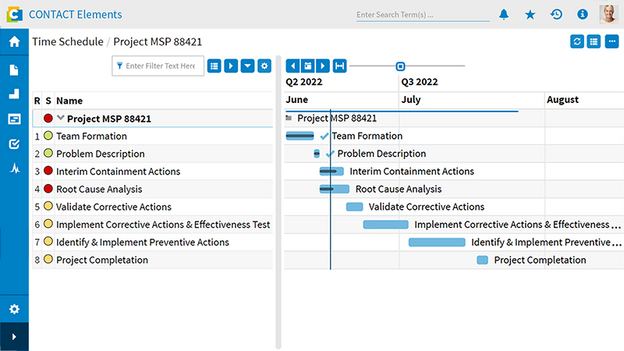
With CONTACT Elements Quality Issue Management, you handle quality defects with system support throughout the entire product lifecycle: from initial inspection through production ramp-up to ongoing production. This always occurs within the relevant product or project context. The solution ensures full conformity with important quality standards such as EN ISO 9001 and ISO 16949 through seamless documentation and traceability of every defect recording and resolution. Thanks to targeted defect resolution, you benefit from higher customer satisfaction, improved product quality, reduced production ramp-up costs, and faster production maturity.
Integrated processes from error recording to the solution archive
Establish a proactive, automated process for defect recording and resolution. Standardized workflows represent your proven solution strategies and best practices. By efficiently recording and evaluating errors, you accelerate their correction. Immediate access to checklists, tickets, and test reports, as well as links between defects and actions, support contextual processing. Comprehensive analysis capabilities and an intelligent solution archive make it easier to learn from mistakes sustainably and continuously improve processes. Seamless integration with systems like CAD or ERP, as well as master data, makes our Quality Issue Management an indispensable foundation for your entire quality and defect management.
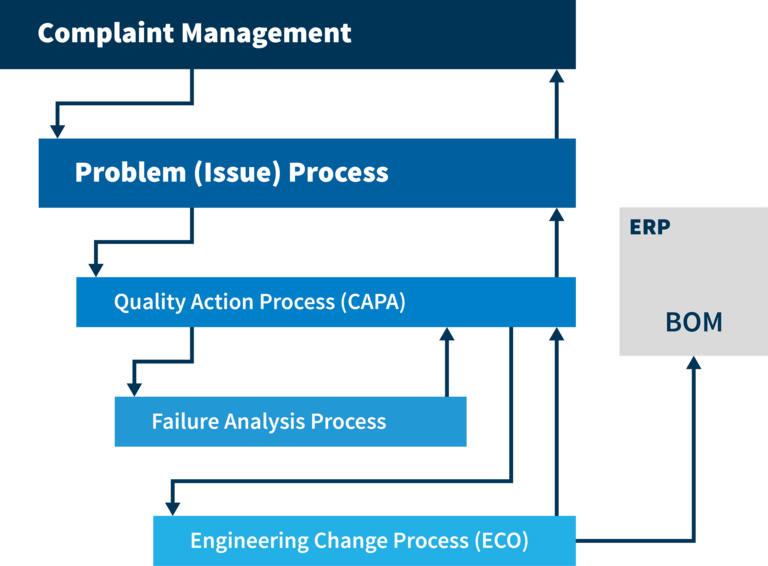
Establish a proactive, automated process for defect recording and resolution. Standardized workflows represent your proven solution strategies and best practices. By efficiently recording and evaluating errors, you accelerate their correction. Immediate access to checklists, tickets, and test reports, as well as links between defects and actions, support contextual processing. Comprehensive analysis capabilities and an intelligent solution archive make it easier to learn from mistakes sustainably and continuously improve processes. Seamless integration with systems like CAD or ERP, as well as master data, makes our Quality Issue Management an indispensable foundation for your entire quality and defect management.
Related Elements
Meet requirements with ease
Document best practices in a digital library
Use proven methods and sets of guidelines such as CAPA, 8D or APQP
Give your employees a way to apply process-related knowledge easily and reliably
Secure projects through proactive risk management
Make risks visible and control them
Meet risk and quality management standards with less effort

